News
Early Clues to Parkinson’s Disease Found in the Gut
Researchers have identified changes in gut bacteria that may signal the earliest stages of Parkinson’s disease in people who carry a genetic risk factor linked to the condition.
In a study published in Nature Medicine, scientists analysed stool samples and clinical data from people with Parkinson’s disease, healthy individuals, and symptom-free carriers of a variant of the GBA1 gene, which increases the risk of developing the disease.
The team found that more than a quarter of gut microbial species differed between healthy people and those with Parkinson’s disease. Strikingly, many of the same bacterial changes were also present in GBA1 carriers who had not yet developed symptoms. Their gut microbiomes appeared to sit midway between those of healthy individuals and people with Parkinson’s disease, and the extent of these changes was linked to subtle early symptoms.
Parkinson’s disease is usually diagnosed only after significant loss of brain cells has occurred. The discovery that gut microbiome changes may appear before noticeable symptoms raises the possibility of identifying people at higher risk earlier.
However, the researchers caution that the study cannot show whether these microbial changes predict future disease. Long-term studies are needed to determine their role in Parkinson’s development.
Original article: Menozzi, E., Ren, Y., Geiger, M. et al. Microbiome signature of Parkinson’s disease in healthy and genetically at-risk individuals. Nat Med (2026). https://doi.org/10.1038/s41591-026-04318-5
Could a new ultrasound technique replace colonoscopy for Crohn's disease monitoring?
Colonoscopy is the current gold standard for checking whether Crohn's disease is active or in remission, but it's invasive, uncomfortable, and can't be done repeatedly. While ultrasound techniques exist, they have limitations in accurately detecting disease activity. A recent study by Wang et al. published in Insights into Imaging (2026) tested a new ultrasound technique called super-resolution contrast-enhanced ultrasound (SR-CEUS) as a potential non-invasive alternative. The technique involves injecting tiny harmless gas bubbles into the bloodstream and tracking them individually through the smallest blood vessels in the gut wall, generating detailed maps of blood flow and vessel structure. In active Crohn's disease, these vessels appear disorganised and tortuous, which is characteristic of inflammation-driven changes in the bowel wall. In a study of 52 patients, SR-CEUS performed significantly better than conventional ultrasound methods at distinguishing active from inactive disease. When combined with standard bowel ultrasound, which assesses structural changes in the bowel wall, it detected every case of active disease, suggesting this combination could be a promising non-invasive monitoring tool for Crohn's patients in the future.
Wang, Y., Ge, W., Yu, Y. et al. A novel super-resolution contrast-enhanced ultrasound approach for evaluating inflammatory activity in Crohn’s disease. Insights Imaging 17, 135 (2026). https://doi.org/10.1186/s13244-026-02309-1
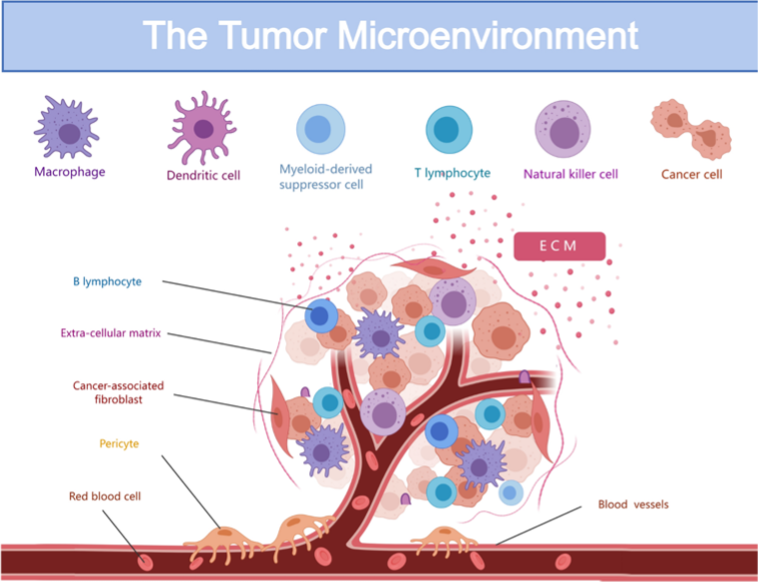
The Myeloid Makeover: Turning Resistance into Response.
The immune response is one of the most important factors that determines how patients respond to cancer. Immunotherapies represent an exciting opportunity to reinvigorate a patient’s immune system to destroy cancer cells. However, solid tumours such as colorectal cancer show variable outcomes, likely due to complex cell interactions within the tumour microenvironment (area within and surrounding the tumour).
Anti-PD-1 monoclonal antibody therapy aims to block the Programmed Death pathway of CD8+ T cells within the tumour and promote the anti-tumour immune response, however myeloid cells such as macrophages and myeloid-derived suppressor cells can promote a continually immunosuppressive environment and therefore promote tumour growth.
A recent study conducted by Mestrallet et al. 2026 has demonstrated that by reprogramming immune cell interactions within the tumour they could overcome immune resistance. The authors targeted TREM2+ macrophages using a combination of anti-LAG3, anti-CTLA-4 and anti-PD-1 to reprogram the tumour microenvironment of BALB/c mice and achieve up to 100% tumour clearance in mismatch-repair deficient cancers and >70% win mismatch repair-proficient models.
This study represents an emerging avenue of immunotherapies and highlights the role of myeloid cells in the success or failure of immunotherapy in colorectal cancer.
For more information:
https://www.cell.com/cell-reports-medicine/fulltext/S2666-3791(26)00203-X
Image credit:
https://link.springer.com/article/10.1007/s12094-024-03697-w

Bread and IBD: Could your sandwich be making you sick?
A study published by Shakman et al in 2025 shone a light on how the ingredients used to make industrial bread, with varying levels of processing, may contribute to inflammation for people with IBD. The authors screened breads available in supermarkets and health food stores, which were then categorised into: low processed (traditional ingredients), medium processed (contained additives such as fibres and malt), and highly processed (contained additives such as emulsifiers and preservatives). They then did a literature review to ascertain the link between these specific additives and inflammation for people with IBD.
The research team screened 233 breads, of which 12% were low processed, 4% medium processed, and 84% highly processed. They identified 37 different food additives and ingredients used, with 81% of breads containing preservatives and 76% emulsifiers. Their literature review showed that 51% of the food additives used in the bread industry were known to either increase gut inflammation or disrupt the gut microbiome.
These findings emphasise the importance of dietary factors, and consideration should be given to recommending low-processed breads to people with IBD.
Article source:
https://doi.org/10.3390/nu17132120
Image source: freepngimg.com

World IBD Day 2026!
Today is World IBD Day!
This year’s theme is Access to Care: The Case for Equitable IBD Care.
Millions of people live with IBD worldwide, yet access to therapies and support still depends too often on geography, resources and health system capacity. Many also continue to face delayed or missed diagnosis, limited access to care, high costs, and gaps in psychosocial support.
Access to IBD care is not a privilege – it’s a right. Quality care should never depend on where someone lives.
Learn more about the campaign here: https://buff.ly/MsygfFs
#IBDHasNoBorders #WorldIBDDay2026 #AccessToIBDCare
Art Credits: crohnsandcolitisnz
Colon the Shots: Letting the Immune System Take Control
Colorectal cancer has traditionally been treated with surgery and chemotherapy. But in 2026, instead of removing the tumour first and treating later, doctors are now training the immune system to attack the cancer before surgery. In effect, the treatment puts up a ‘wanted poster’ for the cancer, helping the immune system to recognise and target it before doctors step in.
A recent study published by AACR (American Association for Cancer Research) showed some quite striking results. 59% of Patients who received an immunotherapeutic drug (pembrolizumab) pre-operation were found to have no detectable cancer by the time of surgery. What makes this even more impressive is that there is no recurrence after three years (so far).
Photo Credit: https://aucklandradiationoncology.co.nz/understanding/bowel-colon-cancer/

Black Liquorice Compound Shows Promise for Treating IBD:
A new study from the University of Tokyo has identified a natural compound found in black liquorice, called glycyrrhizin, as a promising new treatment for inflammatory bowel diseases (IBD). Using a stem cell model of the human intestine, the research team screened 3,500 compounds for their ability to protect gut cells from IBD-related inflammation. Glycyrrhizin stood out from the rest, reducing cell death and inflammation both in the lab model and in mice with IBD.
To simulate the disease, the team exposed the intestinal cells to a protein known to trigger inflammation in IBD patients, causing the cells to become inflamed and begin to die in a way that closely resembles what occurs in the intestines of real patients. Glycyrrhizin was able to protect the cells against this process. These results were then confirmed in a mouse model of IBD, where animals showed less inflammation and fewer damaged cells.
While Glycyrrhizin has been studied before, this research provides some of the strongest evidence yet of its protective effects on gut tissue. Clinical trials in humans are still needed to confirm safety and dosage, but the findings offer an exciting new direction for patients who do not respond well to existing treatments. This research also raises the possibility that a natural compound could one day support or even replace traditional therapies.
Credit: curtoicurto/istock

Can a Fish-Based Diet and Aspirin Help Prevent Colon Cancer?
Diet has been associated with the development and progression of colorectal cancer (CRC) in many studies, often focussed on fibre consumption and limiting saturated fats. This study assessed the chemo-preventative potential of a pesco-vegetarian diet consisting of fish and plant foods in combination with two non-steroidal anti-inflammatory drugs (NSAIDs), acetylsalicylic acid (Aspirin) and Sulindac, against CRC.
PIRC rats (a common CRC animal model) were used in this study, and they assessed tumour burden, inflammatory markers and gut microbiota among other relevant markers to determine the effect of treatment.
They found that the pesco-vegetarian diet reduced total tumours and colon tumours regardless of drug treatment, indicating that this diet alone is protective in the context of CRC. Aspirin showed a dose-dependent positive effect where it reduced tumour incidence. When Aspirin was combined with the pesco-vegetarian diet, an additive benefit was seen, indicating that the diet and drug work synergistically to reduce tumour burden. No significant results were seen regarding Sulindac. Both the pesco-vegetarian diet and Aspirin modulated the gut microbiota towards species linked with gut health, with diet being the strongest driver of microbiota changes.
This study highlights the potential positive effects of NSAIDs in CRC, and how their benefit could be enhanced using a dietary intervention such as the pesco-vegetarian diet. It also suggests microbiota may be part of the mechanism of protection; however, this is preliminary and would need further investigation.
Flagellin Discovery May Predict Crohn's Disease:
A new study, led by Dr. Ken Croitoru at Sinai Health has identified a new biomarker to detect Crohn’s disease (CD) before symptoms begin. Currently CD is diagnosis relies on symptoms and tissue change, which means treatment occurs after damage has already been caused. This new research focuses on early identification of CD before the disease has fully developed.
The team discovered that people who showed a heightened immune response (elevated antibodies) to flagellin, a protein found in the tail-like structures of certain bacteria, were more likely to later develop CD. The identified heightened immune response was targeted specifically to the flagellin of the Lachnospiraceae family, which is bacteria that is thought to be beneficial in a regular intestinal environment.
The study was able to occur using the GEM project, which has investigated 5,000 healthy first-degree relatives of people with CD, of which 130 participants have gone on to develop CD. On average participants were diagnose with CD about 2 and half years after these heightened immune responses to flagellin were detected.
These findings not only suggest that the immune system may play a role in the triggering the disease (rather than reacting to it) but also identify a new biomarker for CD. The development of a biomarker blood test could pave the way for identifying individuals at risk earlier, opening the door to prevention strategies and more proactive care.
Source: https://www.sciencedirect.com/science/article/pii/S1542356525010316?via%3Dihub
Credit: Getty Images/iStockphoto

Can Aspirin Prevent Bowel Cancer?
This clinical trial looked at whether a low dose of aspirin could help prevent bowel cancer from coming back after people had surgery for stage 1–3 tumours with specific mutations. Participants were grouped according to the tumour mutations present. Group A was the primary focus of the trial and included people who had a tumour with a common and specific mutation in the PIK3CA gene. Group B included people who had tumours with other mutations in the PIK3CA, PIK3R1, and PTEN genes. These genes are involved in the PI3K pathway - a cell signalling pathway that helps regulate cell growth. When faulty, it can contribute to uncontrolled cell division and tumour growth.
A total of 626 participants from four Nordic countries took part. Within 12 weeks of surgery, half of the participants received daily aspirin and half received a placebo, in addition to standard clinical treatment. After 3 years of follow-up, participants in Group A who took aspirin had a significantly lower rate of cancer recurrence (7.7%) compared to those who took the placebo (14.1%). A similar benefit was seen in Group B, though this was a secondary finding. Serious side effects were uncommon but slightly more frequent in the aspirin group compared to the placebo group. This trial provides evidence that taking aspirin may help reduce cancer recurrence in patients with certain tumour mutations. While more research is needed to determine the best dose and duration, and whether factors like weight and sex affect outcomes, aspirin could be a cost-effective and accessible additional treatment for bowel cancer.

Is the weight loss drug semaglutide (Wegovy/Ozempic) safe for people with inflammatory bowel diseases?
Semaglutide (branded as Wegovy/Ozempic) is a medication that has recently been approved for use in Aotearoa New Zealand as a weight loss treatment. It is a GLP-1 (glucagon-like-peptide-1) receptor agonist and was originally developed and utilised to manage type 2 diabetes. Semaglutide has also been shown to be effective for weight loss. It functions by binding to GLP-1 receptors, resulting in slowed gastric emptying and reduced appetite but it can also cause gastrointestinal adverse events in some people. This raises the question, is semaglutide safe for people with pre-existing gastrointestinal complications, such as those with inflammatory bowel diseases (IBD)?
Two recently published papers in the journal of Inflammatory Bowel Diseases sought to answer this question. First, to determine whether semaglutide was safe for those with IBD and second, whether semaglutide remained effective for weight loss in people with IBD.
The first study by Levine et al. showed that IBD patients taking semaglutide did not experience increased adverse IBD-related complications compared to those not taking the drug. The second paper by Desai et al. confirmed this result and further demonstrated that semaglutide remained as effective for weight-loss drug in obese people with IBD compared to a non-IBD obese population also taking the drug.
Together, these studies indicate that semaglutide is safe and effective for those with IBD, suggesting that using this treatment for co-morbidity with obesity could be considered without increased risk of IBD-related disease flares. However, it is always important to speak with your doctor before deciding whether a new medication is right for you.
To read about these studies in more detail follow the links below:
https://doi.org/10.1093/ibd/izae250
